TAMPA, Fla. — Researchers, scientists, and health professionals all over the world are working around the clock to find treatments for the coronavirus. There are several trials going on right now, but it could take months to find which ones work best.
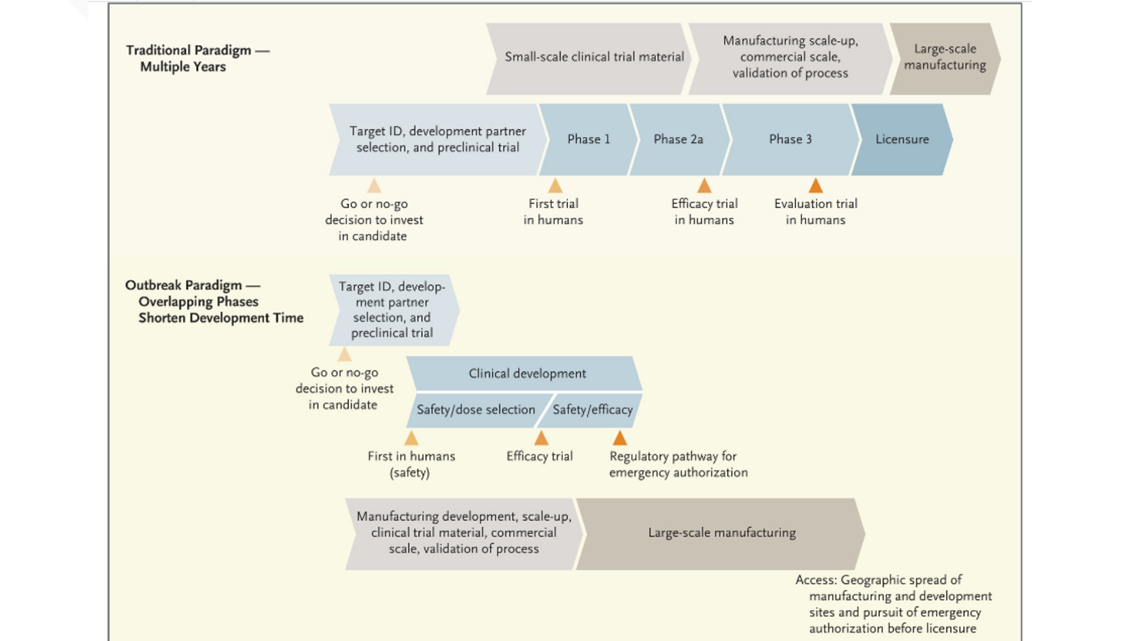
They are also working to find a vaccine. But according to the New England Journal of Medicine that could take years.
Developing a vaccine takes time and is an expensive process. It is going to take multiple candidates and years to produce a licensed vaccine.

In the meantime, let's focus on some of the treatments currently on trial or being tested.
Remdesivir-- is currently at the top of the list of treatments the world is excited about.
On May 1, 2020, the Food and Drug Administration (FDA) allowed the emergency use of Remdesivir as a treatment for COVID-19. The experimental drug appears to help some coronavirus patients recover faster.
The research-based biopharmaceutical company, Gilead Sciences, Inc. has been studying Remdesivir for more than 10 years.
Gilead scientists tested the drug as a treatment for hemorrhagic fever viruses such as Ebola, Marburg, and Nipah viruses, as well as other coronaviruses such as SARS and MERS.
When COVID-19 started to turn into a global pandemic, Gilead Sciences put Remdesivir into clinical trials to test how it would work as a treatment. The National Institute of Allergy and Infectious Diseases sponsored the Adaptive COVID-19 Treatment Trial, ACTT.
In February 2020, the first study site opened at the University of Nebraska Medical Center. The trial stopped taking new volunteers on April 19.
One of the ACTT trial sites is right here in the Tampa Bay area at Sarasota Memorial Hospital (SMH.)
There are eight people enrolled in the ACTT trial at SMH. "We are using the experimental drug in very sick people on ventilators, so it is difficult to assess the benefit of the drug on our patients at this time,” said Kirk Voelker, a critical care pulmonologist caring for COVID-19 patients in Sarasota Memorial Hospital’s ICU and medical director and sub-investigator of the health system’s COVID-19 research trials.
Although SMH is in the early phase of the ACTT trial, Voelker said things look promising. "We are seeing positive indications in the trial and are hopeful that this treatment, along with other therapies we are researching, will be effective.”
The initial group of people in the ACTT trial are Americans who quarantined aboard the Diamond Princess cruise ship, which was docked in Yokohama, Japan.
There are 68 study sites around the world, including 47 in the United States. The other test sites are located throughout Europe and Asia.
This week, Dr. Anthony Fauci, director of the NIAID, said findings from the Remdesivir trial are promising. He said the drug reduced the time it takes people to recover by 31 percent — 11 days on average versus 15 days for those just given usual care.
“What it has proven is that a drug can block this virus. This will be the standard of care," Dr. Fauci said.
The National Institutes of Health (NIH) also followed up with its own study and found patients who took Remdesivir recovered faster than patients who did not.
However, the NIH study also found eight percent of patients who took the drug died versus 11.6 percent of patients who did not take Remdesivir.
Chloroquine – This drug is primarily used to prevent or treat malaria, which is caused by mosquito bites.
It’s currently in clinical trials. And as with any drug, researchers realized chloroquine is not ideal for every patient.
Research of how effective chloroquine is for COVID-19 is still in the beginning stages. And collecting enough data might take longer than anticipated because currently there’s a shortage of the medication.
The FDA put out warning labels for users of Chloroquine Phosphate.
They list cardiac effects, hypoglycemia, retinopathy, central nervous system effects, muscular weakness, and potential carcinogenic risk as possible side effects.
A report from the Institute of Virology of the Chinese Academy of Sciences showed the dosing of Chloroquine is very sensitive and taking too much could have major side effects or even death.
The FDA has yet to approve chloroquine as a treatment for COVID-19
Hydroxychloroquine – is less toxic than chloroquine.
It is prescribed to those with rheumatoid arthritis, lupus, and porphyria cutanea tarda, a blood disorder that affects 1 out of 25,000 people.
Hydroxychloroquine is being tested as a preventative for healthcare workers. If it works for health professionals, will the general population gain access to it as a preventative drug?
Plasma Infusions – Doctors are using plasma infusions on COVID-19 patients on ventilators.
Blood banks and the American Red Cross are asking for plasma donations from people who have tested positive for COVID19 and then negative. Donors should be symptom-free for 28 days.
According to the American Society of Hematology, convalescent plasma could provide short-term immunity against the SARS-CoV-2 coronavirus.
Most patients who recover from COVID-19 develop antibodies to various SARS-CoV-2 proteins two to three weeks after being infected. Transfer of plasma from these patients is supposed to neutralize the virus and stop tissue damage.
Plasma infusions should work best on those with less severe infection, according to the American Society of Hematology.
Azithromycin – Is an antibiotic that has never been used for viral infections. According to the U.S. National Library of Medicine, Azithromycin is used to treat bacterial infections, such as bronchitis, pneumonia, sexually transmitted diseases and infections of the ears, lungs, sinuses, skin, throat, and reproductive organs.
So far, there hasn’t been any solid research, but some medical observers found that azithromycin could help reduce an overactive immune response to the COVID-19 infection. – Harvard Medical School
Studies that have been done suggest azithromycin can be deadly if it’s used in combination with hydroxychloroquine. The human heart just couldn’t hold up.
Vitamin C -- Some people who are critically ill with COVID-19 have been treated with high doses of vitamin C through their IV. Doctors hope high doses of vitamin C will speed up the recovery process.
There’s no scientific evidence that a vitamin C IV drip works for COVID-19 infections.
Researchers in China are currently conducting a study to see if the high doses of vitamin C will help those with severe COVID-19. Results are expected in the fall of 2020. – Harvard Medical School
Lopinavir–ritonavir – is an antiviral drug used to treat patients with human immunodeficiency virus, HIV. The drug is currently on the Chinese clinical trial register and is administered to adults with severe COVID-19 symptoms.
So far, doctors have not seen any benefits of the drug as a treatment.
What are we missing?
As mentioned at the beginning of this article, everyone is chipping in to find an ideal treatment for COVID-19.
What treatments or clinical trials have you learned about?
E-mail your findings to TCody@wtsp.com and I'll dig into the research and then ask the experts how well the treatments are working.
What other people are reading right now:
- Gov. DeSantis says reopening can begin May 4, restaurants can open with restrictions
- Where's the bacon? We're 'about 3 weeks away from not having pork on the shelves,' congressman says
- 100 bodies found decomposing in rental trucks outside New York funeral home
- 'Ineligible' for unemployment assistance? Florida wants you to apply again
- Pinellas County beaches reopening Monday with restrictions
- 10Investigates: Renters faced with rent hikes during pandemic
- Hotlines, websites offer the latest on COVID-19